Link to:
Summary
Dr David Fengwei Xie possesses a wealth of research experience at the crossroads of engineering, chemistry, and physics, with a particular focus on biopolymers (polysaccharides and proteins) for ‘green’ materials and food applications. He has honed his expertise extensively in various biopolymers, including starch, chitosan, cellulose, alginate, and gelatin. His research endeavours have been dedicated to exploring the following aspects of biopolymers:
- Multilevel structures
- Molecular interactions (e.g. hydrogen bonding, ionic interaction)
- Dissolution, plasticisation and gelation
- Chemical and physical modifications
- Polymer processing and materials engineering (e.g. reactive processes, sustainable engineering)
- Structural evolution during processing, modification, ageing and usage
- Blends and (nano)composites
- Processing-structure-property relationships
Featured research findings
Starch and ionic liquid
The thermal transition of starch is largely influenced by ionic liquid (IL)/water ratio. Aqueous IL with a certain IL/water ratio leads to the most effective structural disorganisation and amorphisation of starch at significantly reduced temperature (even at room temperature) (Carbohydr. Polym. 2013, 94, 520-530; Phys Chem Chem Phys 2015, 17, 13860-13871; ACS Sustainable Chem. Eng., 2017, 5 (5), 3737-3741), a phenomenon very different from the dissolution of cellulose in ILs. Regarding this, the surface pores on starch granules (e.g. cassava and waxy maize) allow the corrosion by the aqueous IL to follow an inside-out pattern, making granule destruction easy and fast (Carbohydr. Polym. 2021, 258, 117677). While starches with a smooth granule surface (e.g. potato and high-amylose maize) can hardly be structurally disrupted simply by the aqueous IL, they become processable at room temperature once mechanically fractured (Sustainable Mater.Technol. 2023, 36, e00592).
The use of such IL:water mixtures can enable the effective plasticisation of starch in a highly concentrated state under “melt” processing at a moderate temperature (≤65 °C) (ACS Sustainable Chem. Eng. 2017, 5 (6), 5457-5467).

Starch and metal chloride salts
Starch, even high-amylose starch, can be fully dissolved by aqueous metal chloride salts (e.g. ZnCl2, CaCl2, MgCl2) at a moderate temperature (≤50 °C); starch nanoparticles are formed during this dissolution process (Carbohydr. Polym. 2016, 136, 266-273; ACS Sustainable Chem. Eng. 2020, 8 (12), 4838-4847). Under “melt” processing, ZnCl2 solution has an excellent plasticisation effect on starch, and in-situ formed starch-zinc complexes can enhance the mechanical properties of starch-based materials (Carbohydr. Polym. 2019, 206, 528-538). Simply mixing CaCl2 solution with starch can lead to starch-based materials with ionic conductivity and strain-responsiveness (ACS Sustainable Chem. Eng. 2020, 8 (51), 19117-19128). Based on starch/CaCl2/glycerol hydrogel, flexible electronics including strain-sensitive batteries and self-powered wearable sensors can be constructed (ACS Sustainable Chem. Eng. 2022, 10, 20, 6724-6735). Starch/Ca2+ gel can also be used as a flame-retardant adhesive for paper and wood board (Molecules 2023, 28 (11), 4543).

Chitosan-based composites
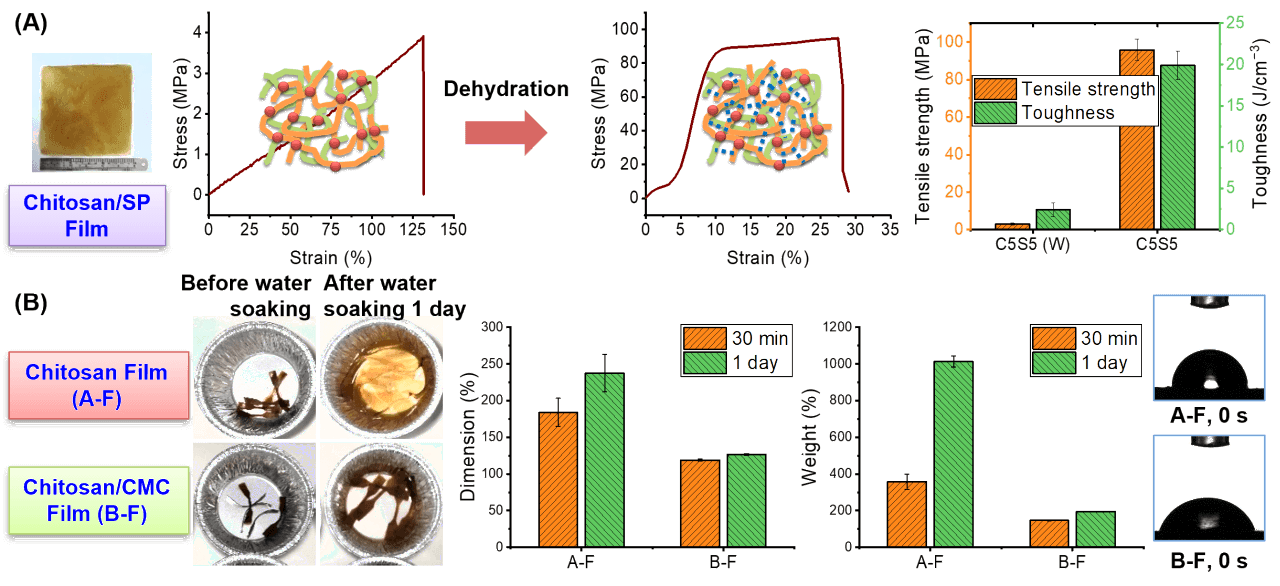
Under “melt” processing with limited solvents, chitosan-based materials and composites can be prepared cost-effectively (Polymer 2013, 54 (14), 3654-3662). Prepared in this way, chitosan blends with other biopolymers (e.g. silk peptide, carboxymethyl cellulose, or gelatin) show extraordinary mechanical properties and unexpected hydrolytic stability, better than that of each biopolymer component (ACS Sustainable Chem. Eng. 2019, 7 (2), 2792-2802 on chitosan/silk peptide, Compos. Sci. Technol. 2020, 189, 108031 on chitosan/carboxymethyl cellulose, and Carbohydr. Polym. 2021, 272, 118522 on chitosan/gelatin), likely due to polyelectrolyte complexation. In these studies, we have also revealed that the overall material hygroscopicity and the surface hydrophilicity are controlled by different mechanisms.
Interesting observations under transmission electron microscopy (TEM)
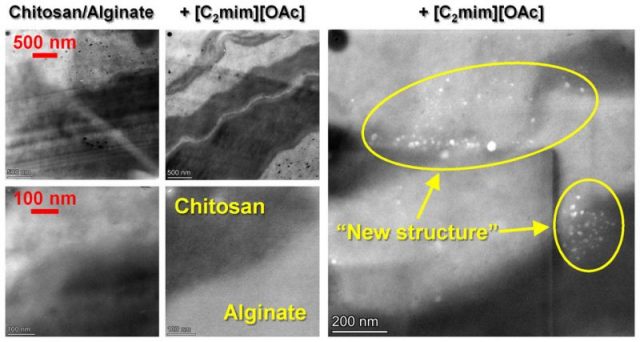
“New structure” formed in biopolymer materials during TEM imaging
Images below: When imaging chitosan/alginate materials using TEM, we found the formation of a “new structure” from biopolymer and ionic liquid under the electron beam (ACS Appl. Polym. Mater. 2020, 2 (7), 2957-2966).
Interconnected structure of nanofillers in biopolymer matrices
Images below: TEM shows interconnected microstructure composed of graphene oxide and sepiolite in chitosan and chitosan/carboxymethyl cellulose matrices, the formation of which could be due to the strong interactions between these hydrophilic nanofillers (Funct. Compos. Mater. 2021, 2, 14).

Research projects (selected)
- “Biopolymer-based functional aerogel materials for tissue engineering applications”, Royal Society, Research Grants 2022 Round 2 (RGS\R2\222071), GB£19,957.54, 10/2022–10/2023
- “Tough, strong natural biopolymer-based hydrogels for artificial muscles”, Royal Society, International Exchanges 2022 Round 1 (IES\R1\221039), GB£11,930, 08/2022–08/2024
- “FROBCO”, EPSRC Fellowship (EP/V002236/1), GB£1,629,558 (fEC), 01/2021–12/2025,
- “ROBBINS”, Marie Skłodowska-Curie Individual Fellowships (798225), 01/2019–12/2020, €195,455
- “Highly functional green materials platform: Starch-ionic liquid-carbon nanotube polymer melt nanocomposites”, ARC Discovery Project (DP120100344), AU$300k, 01/2012–12/2014